
© 1978 American Institute of Physics for the National Institute of Standards and Technology.The two empirical equations are compatible with each other to 0.09%. The less accurate equation covers the wider range −8 ☌? t?150 ☌ with the more limited accuracy of ☐.2%. The more accurate equation covers the range 0 ☌? t ?40 ☌ with an uncertainty of ☐.05%. The viscosity ratios have been correlated with the aid of two empirical equations. The ratios are used to generate absolute values with the aid of the standard NBS datum μ=1002.0 μPa s at 20 ☌. Standard values of the ratio of viscosity at a temperature T to its value at 20 ☌ have been derived from the re‐analyzed data because the uncertainty of this ratio is an order of magnitude smaller than that of the absolute values. The latter are smaller than those embodied in the most recent International Formulation. 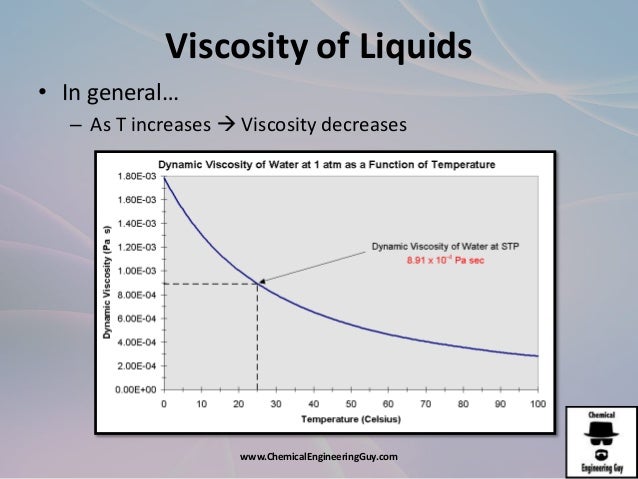
The new analysis eliminates possible systematic errors and permits the establishment of realistic error bounds for water in its role as a standard reference substance for viscosity. This is done in terms of a new, theoretically‐based equation for the operation of a capillary viscometer rather than in terms of semi‐empirical equations used by the original authors. The paper re‐analyzes the results of earlier, very precise measurements of the viscosity of water at essentially atmospheric pressure. The increase in viscosity for sucrose solutions is particularly dramatic, and explains in part the common experience of sugar water being 'sticky'.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
